Characterizing the immunobiology of high risk pediatric ependymoma
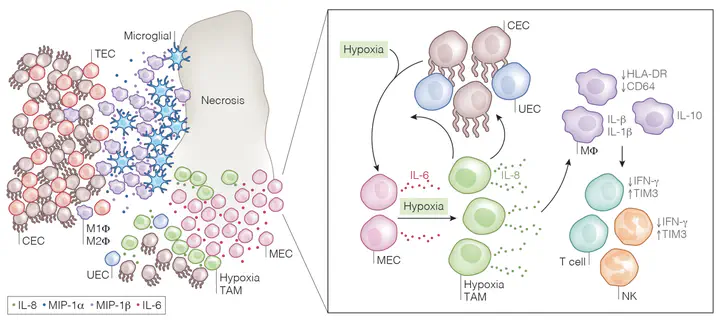
Ependymoma (EPN) is a devastating childhood brain tumor. Single-cell analyses have illustrated the cellular heterogeneity of EPN tumors, identifying multiple neoplastic cell states including a mesenchymally-differentiated subpopulation which characterizes the PFA1 subtype. Here, we characterize the EPN immune environment, in the context of both tumor subtypes and tumor cell subpopulations using single-cell sequencing (scRNAseq, n=27), deconvolution of bulk tumor gene expression (n=299), spatial proteomics (n=54), and single-cell cytokine release assays (n=12). We identify eight distinct myeloid-derived subpopulations from which a group of cells, termed hypoxia myeloid cells, demonstrate features of myeloid-derived suppressor cells, including IL6/STAT3 pathway activation and wound healing ontologies. Hypoxia myeloid cells colocalized with mesenchymally-differentiated cells in necrotic and perivascular niches in PFA tumors and secrete IL-8, which we hypothesize amplifies the EPN immunosuppressive microenvironment. Childhood EPN immunotherapy must overcome the myeloid cell driven immunosuppressive tumor microenvironment and our data provides new routes for therapeutic innovation in this difficult to treat disease.
Accompanies the manuscript by Griesinger et al. “Multi-Omic approach identifies hypoxic tumor-associated myeloid cells that drive immunobiology of high risk pediatric ependymoma”
Data availability
snRNA-seq was previously published and is available in the National Center for Biotechnology Information Gene Expression Omnibus (GEO) database at accession GSE126025.
A UCSC cellbrowser is available for interactive exploration of the data.
Supported by
National Institutes of Health (R01 CA237608-01)
Cancer League of Colorado (research grant AWD 173796-SV)
Tanner Seebaum Foundation
Morgan Adams Foundation
University of Colorado’s NIH/NCI Cancer Center (P30CA046934)